AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Violation aufbau principle8/15/2023 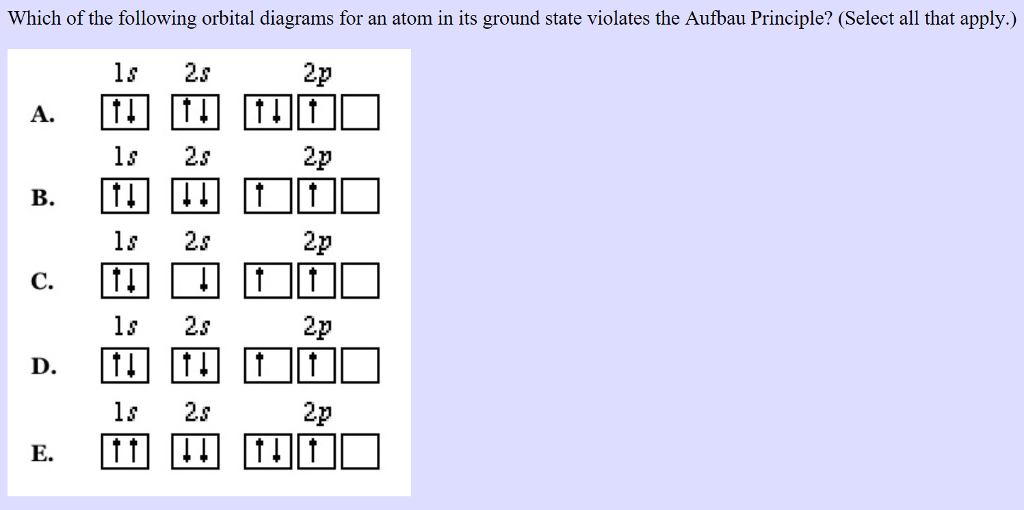
Madelung energy ordering rule įor multielectron atoms the energy spectra of shells interleave resulting in the n + l rule For subshells with the same value of n + l, electrons are assigned first to the subshell with lower n.Ī version of the aufbau principle known as the nuclear shell model is used to predict the configuration of protons and neutrons in an atomic nucleus.Electrons are assigned to subshells in order of increasing value of n + l.Thus subshells are filled in the order of increasing energy, using two general rules to help predict electronic configurations: In the ground state, the electronic configuration can be built up by placing electrons in the lowest available subshell until the total number of electrons added is equal to the atomic number. The maximum number of electrons in a subshell is equal to 2(2 l + 1), where the azimuthal quantum number l is equal to 0, 1, 2, and 3 for s, p, d, and f subshells, so that the maximum numbers of electrons are 2, 6, 10, and 14 respectively. The maximum number of electrons in any shell is 2 n 2, where n is the principal quantum number. 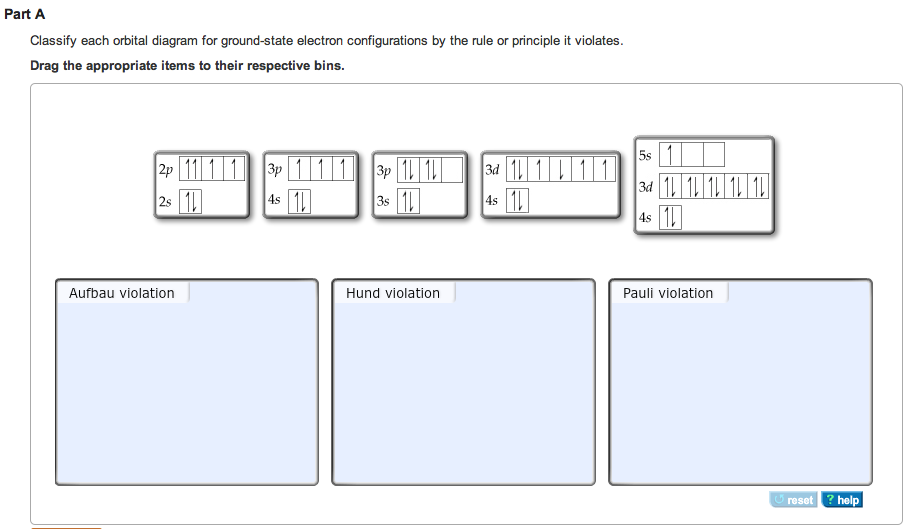
Passing from one element to another of the next higher atomic number, one proton and one electron are added each time to the neutral atom. If double occupation does occur, the Pauli exclusion principle requires that electrons that occupy the same orbital must have different spins (+ 1⁄ 2 and − 1⁄ 2). Hund's rule asserts that if multiple orbitals of the same energy are available, electrons will occupy different orbitals singly and with the same spin before any are occupied doubly. An example is the configuration 1s 2 2s 2 2p 6 3s 2 3p 3 for the phosphorus atom, meaning that the 1s subshell has 2 electrons, and so on.Įlectron behavior is elaborated by other principles of atomic physics, such as Hund's rule and the Pauli exclusion principle. In this way, the electrons of an atom or ion form the most stable electron configuration possible. For example, the 1s subshell is filled before the 2s subshell is occupied. 
The aufbau principle ( / ˈ aʊ f b aʊ/, from the German Aufbauprinzip, which means "building-up principle"), also called the aufbau rule, states that in the ground state of an atom or ion, electrons fill subshells of the lowest available energy, then they fill subshells of higher energy.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
